The U.S. medical community has seen a significant breakthrough: for the first time, cellular therapy has been approved for patients with severe aplastic anemia. This decision caught many experts by surprise, as such methods had previously been used only in clinical trials or for treating other blood disorders. Now, a stem cell-based drug has officially been authorized for use in both children and adults starting at six years old.
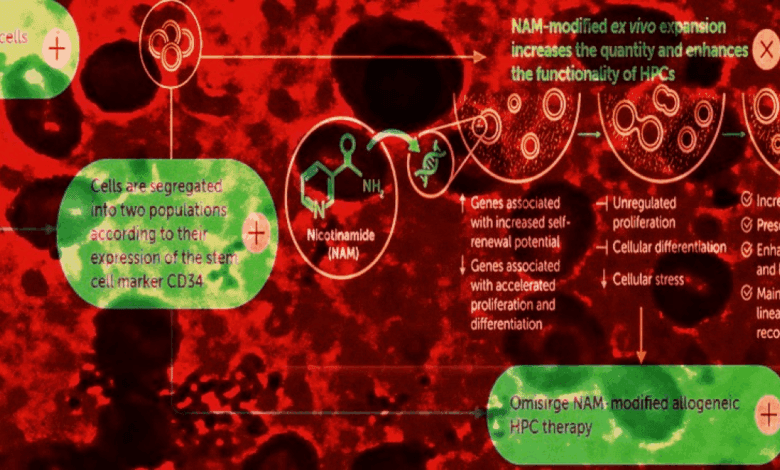
The drug in question has been in development for several years and has already proven effective in treating oncohematological diseases. Its uniqueness lies in the fact that the stem cells are derived from donor umbilical cord blood unrelated to the recipient. Additional processing with nicotinamide allows the cells to retain their properties, accelerating the recovery of blood formation in patients.
New Horizons
For patients with severe aplastic anemia, when the bone marrow nearly stops producing blood cells, the advent of such therapy opens up new possibilities. Until now, the main treatment options were bone marrow transplantation from a compatible donor or prolonged immunosuppressive therapy, which did not always yield the desired results. Now, doctors have an additional tool that can significantly improve the chances of recovery.
Spanish hematologists are closely monitoring developments in the United States, as similar innovations may soon be introduced in European clinics. In recent years, Spain has seen an increase in patients with rare blood disorders, making the adoption of new treatment methods increasingly relevant. Doctors note that the use of donor stem cells from umbilical cord blood could be a real breakthrough for those who previously had no access to suitable transplants.
Technology and capabilities
The key feature of the approved drug is its ability to rapidly engraft in the recipient’s body. Thanks to special processing, the stem cells not only retain their viability but also integrate more actively into the process of blood formation. This is especially important for patients with severe aplastic anemia, for whom time is of the essence.
In clinical trials, the drug demonstrated high efficacy and an acceptable safety profile. Patients who received the new therapy saw their blood counts recover more quickly and experienced fewer complications related to transplant rejection. Doctors emphasize that such results offer hope for improving the quality of life for thousands of people suffering from this rare and dangerous disease.
Looking ahead
The approval of cell therapy in the United States could mark a turning point in the global standards for treating aplastic anemia. There is already discussion around expanding the indications for this drug and integrating it into protocols for other blood disorders. In Spain, experts expect that in the coming years, similar technologies will become available to local patients, which could dramatically improve treatment outcomes.
The medical community notes that the arrival of new treatment methods always raises questions about accessibility and cost. However, with cell technologies, specialists believe that the benefits for patients and the healthcare system as a whole could outweigh the potential expenses. New data on the long-term outcomes of this therapy is expected to be published soon, allowing for a more accurate assessment of its potential.
Reaction in Spain
Spanish patients and their families are looking to the news from the United States with hope. Many face limited treatment options and are forced to seek care abroad. The approval of innovative therapy in America could speed up its registration process in Europe and, in particular, in Spain. Doctors are already preparing for the possible adoption of new protocols and undergoing training to work with cell-based therapies.
Within professional circles, the discussion extends beyond the medical aspects to include ethical considerations as well. The use of donor umbilical cord blood requires strict adherence to safety and ethical standards. Nevertheless, most experts agree that the benefits of the new therapy are evident and could change the approach to treating severe blood disorders.
If you didn’t know, the FDA (Food and Drug Administration) is the American agency responsible for regulating the quality of medicines and medical technologies. This organization makes decisions about the approval of new drugs for the U.S. market. The approval of cell therapy for the treatment of aplastic anemia has become one of the most talked-about events in the medical world in 2025. The drug Omisirge is already being used in a number of clinics for the treatment of other blood disorders and is considered one of the most promising in its field.












