A debate has erupted in European medical circles following the publication of phase three clinical trial data on a new antibiotic capable of curing uncomplicated gonorrhea with just a single dose. Spanish experts are closely monitoring developments, as resistance of the pathogen to existing medications has become an increasingly urgent issue. According to WHO estimates, there are more than 80 million new cases of the disease worldwide each year—and Spain is no exception.
Gonorrhea, caused by the bacterium Neisseria gonorrhoeae, has long been considered one of the most insidious sexually transmitted infections. Without prompt treatment, it can lead to serious complications, including pelvic inflammatory disease, joint damage, and even heart problems. In recent years, the gonococcus’s resistance to antibiotics has reached critical levels, forcing physicians to search for alternative therapies and new drugs.
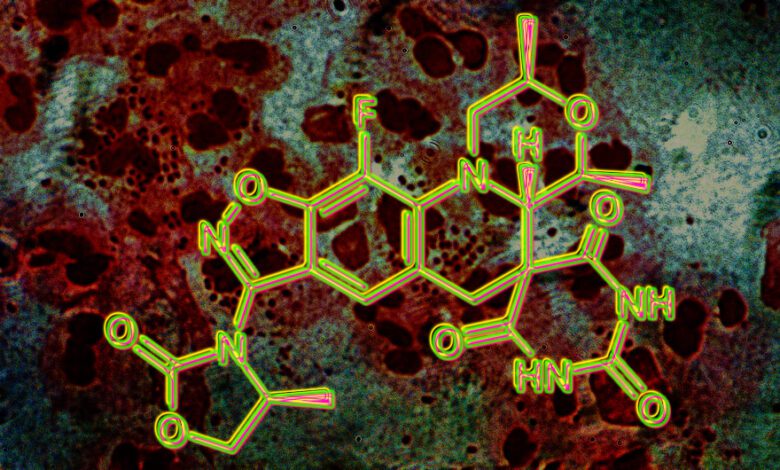
The new molecule
The experimental antibiotic zoliflodacin, developed by an international team with the involvement of Innoviva Specialty Therapeutics and the Global Antibiotic Research & Development Partnership (GARDP), is the first representative of the spiropyrimidinetrione class. Its main advantages are that it can be taken orally and has a pronounced bactericidal effect. The mechanism of action is based on irreversible binding to bacterial DNA gyrase, which disrupts replication and transcription processes, preventing the pathogen from repairing its DNA.
In laboratory settings, zoliflodacin demonstrated activity against strains of gonococcus resistant to ceftriaxone, ciprofloxacin, and azithromycin. Additionally, the drug showed effectiveness against several other pathogens, including staphylococci, streptococci, Haemophilus influenzae, Moraxella, chlamydia, and mycoplasma. This broad spectrum of action has attracted the attention of infectious disease specialists in Spain and other European countries.
Trials in five countries
As part of the phase three clinical trial conducted at 17 outpatient clinics in five countries with high rates of gonorrhea, 930 patients over the age of 12 participated. The average age was 29.7 years, and most participants were men. Volunteers were randomly divided into two groups: one received a single oral dose of three grams of zoliflodacin, while the other received the standard combination of ceftriaxone and azithromycin.
A week after taking the medications, patients were swabbed for microbiological analysis. In the main group, the cure rate was 90.9%, while in the control group it was 96.2%. The difference was statistically insignificant, allowing the new antibiotic to be considered at least as effective as the existing treatment regimen. Importantly, zoliflodacin was well tolerated: side effects were rare and mild or moderate.
Side effects
In the group receiving zoliflodacin, the most common side effects were headache, neutropenia, and leukopenia. Among patients treated with the standard combination, injection site pain, neutropenia, and diarrhea prevailed. No serious complications were reported in either group, which is especially important for the drug’s potential widespread use.
Spanish experts emphasize that the introduction of a new oral antibiotic could significantly change the approach to treating gonorrhea, especially amid rising resistance to cephalosporins. Doctors note the convenience of a single-dose regimen and the absence of injections, which facilitates treatment and improves patient adherence.
Outlook for Spain
The United States Food and Drug Administration (FDA) is already reviewing an application to register zoliflodacin as a new drug. If approved, the medication could enter the European market in the coming years. Spanish clinics are preparing for the possible introduction of this innovative therapy, while pharmaceutical companies are assessing the prospects for production and distribution of the new antibiotic.
Amid a historic peak in gonorrhea cases in the UK, a free vaccination program was launched in August 2025. While there is still no specific vaccine for gonorrhea, the 4CMenB vaccine—originally developed for meningococcus group B—is being used for prevention. Experts estimate it provides about 40% protection against gonorrhea, which is already considered a significant breakthrough.
In case you didn’t know, GARDP (Global Antibiotic Research and Development Partnership) is an international nonprofit organization focused on developing new antibiotics to combat resistant infections. Innoviva Specialty Therapeutics specializes in creating innovative treatments for infectious diseases. Their joint work on zoliflodacin has drawn global attention from the medical community, including in Spain, where the issue of antibiotic resistance is particularly pressing.












